 Example: Transition Elements.į-Block elements are those elements that have their outermost electrons in f-orbitals. These elements produce colored salts and have high melting or boiling points. Example: Carbon, Oxygen, and Sulfur.ĭ-Block elements are those elements that have their outermost electrons in d-orbitals. These elements form covalent bonds by the mutual sharing of electrons. P-Block elements are those elements that have their outermost electrons in p-orbitals. Example: Alkali metals and Alkaline Earth Metals. 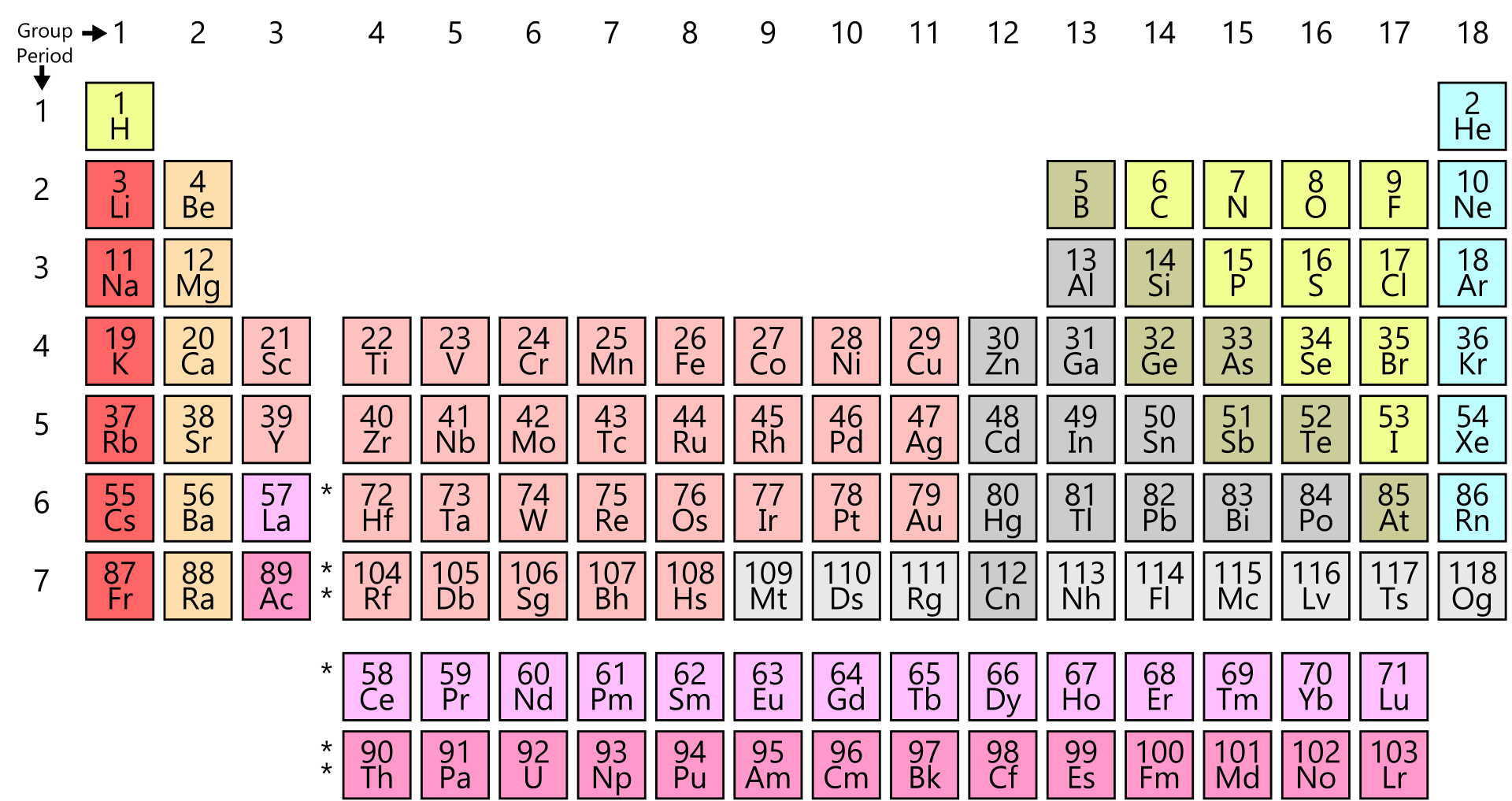
These elements have low melting points, are more electropositive, chemically active, and are soft. S-Block elements are those elements that have their outermost electrons in s-orbitals. → They all are naturally occurring radioactive elements.Īll the chemical elements of the periodic table of elements are arranged in four specific blocks that described the position of the element’s outermost electron in a specific orbital. → They all are f-block elements and inner transition elements.įr, Ra, Ac, Th, Pa, U, Np, Pu, Am, Cm, Bk, Cf, Es, Fm, Md, No, Lr, Rd, Db, Sg, Bh, Hs, Mt, Ds, Rg, Cn, Uut, Fl, Uup, Lv, Uus, and Uuo. → They all have the same general structure and a radioactive element Tc is also included in this period.Ĭs, Ba, La, Ce, Pr, Nd, Pm, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, Lu, Hf, Ta, W, Re, Os, Ir, Pt, Au, Hg, Tl, Pb, Bi, Po, At, and Rn. Rb, Sr, Y, Zr, Nb, Mo, Tc, Ru, Rh, Pd, Ag, Cd, In, Sn, Sn, Te, I, and Xe. /ecblocks-56a129535f9b58b7d0bc9f2e.jpg)
→ They all belong to the first period whose valence shell electrons lie in characteristic d-block orbital thus named as D-block elements. K, Ca, Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Ga, Ge, As, Se, Br, and Kr. → They have electrons in both s and p orbitals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
